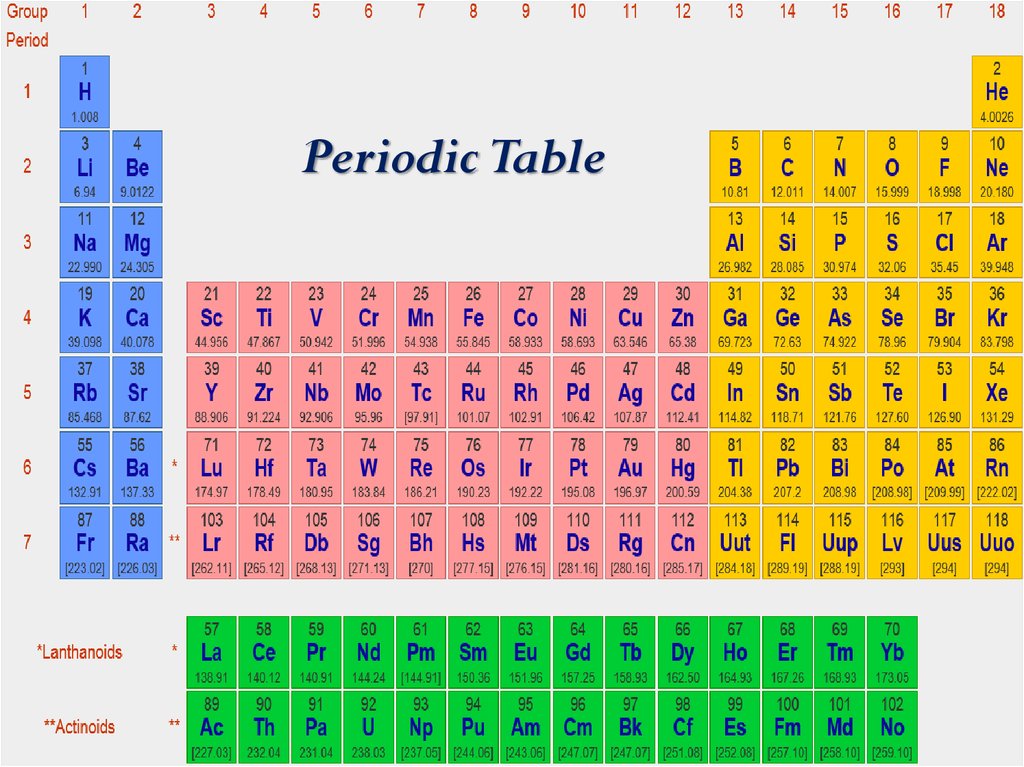 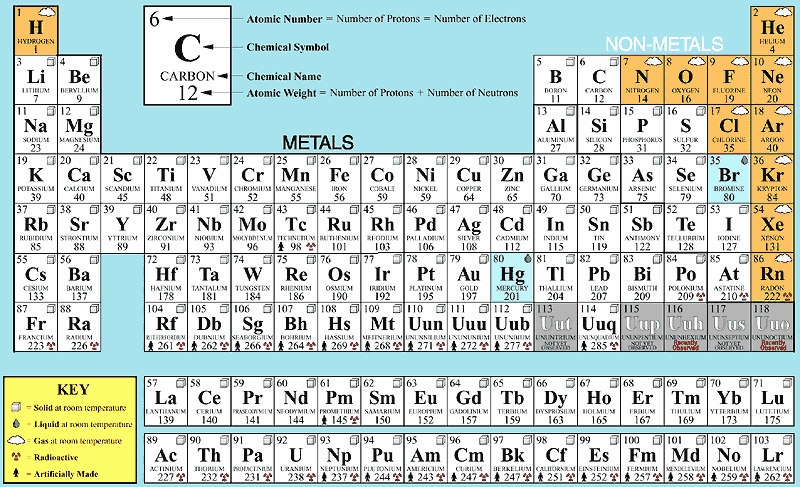
Example: 'Ca' representing Calcium, 'He' representing Helium, etc. The first letter of a symbol is capitalized with the second (or third) letters being in lowercase. Rules or Conventions followed to denote the Element using Symbol The symbol 'Fe' is used to denote Iron, as the Latin word for Iron is "Ferrum". Hence gold is denoted by the symbol ‘Au’. Some symbols of a few elements are derived from their Latin or Greek names. One may ask, ‘How is the symbol of an element derived?’ We can see in the table above that most of the symbols are derived from the elements’ names, by taking either the first or first two letters from the English name of the element. Some symbols have three letters, they generally represent synthesized elements newly, with some being temporarily named like that. Example: Elements in group 1A are soft metals that react violently with water.Ī symbol representing a chemical element is a 'sign' or 'notation' that generally consists of one or two letters. Elements arranged vertically in columns are called ‘Groups’ and elements arranged horizontally in rows are called ‘Periods’.įurther elements are grouped as per periodic trends and properties. The valence electrons are held closer towards the nucleus of the atom.Key Characteristics of the Periodic Table:Įlements are arranged in order of increasing atomic number.Įlements of the Periodic Table are denoted by a unique symbol and not its entire name, as some elements’ names can be long and complex in nature.Įlements are arranged vertically and horizontally. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. 
However, at the same time, protons are being added to the nucleus, making it more positively charged. This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius. Electron affinity decreases from top to bottom within a group.This is caused by the decrease in atomic radius. Electron affinity increases from left to right within a period.This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. \( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
